The Journal Dalton Transactions (IF 4,052) published the article with the participation of NIOCh's researchers :
High molecular weight polyethylenes of narrow dispersity promoted using bis(arylimino)cyclohepta[b]pyridine-cobalt catalysts ortho-substituted with benzhydryl & cycloalkyl groups
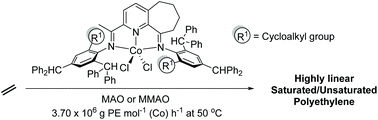
Abstract
A one-pot template strategy has been utilized to synthesize sterically enhanced bis(imino)cyclohepta[b]pyridine-cobalt(II) chlorides, [2-{(Ar)N
CMe}-9-{N(Ar)}C10H10N]CoCl2 (Ar = 2-(C5H9) -4,6-(CHPh2)2C6H2 Co1, 2-(C6H11)-4,6-(CHPh2)2C6H2 Co2, 2-(C8H15)-4,6-(CHPh2) 2C6H2 Co3, 2-(C12H23)-4,6-(CHPh2 )2C6H2 Co4, 2,6-(C5H9)2 -4-(CHPh2)C6H2 Co5). All five complexes have been characterized by a combination of FT-IR spectroscopy, elemental analysis and single crystal X-ray diffraction. The molecular structures of Co1, Co3 and Co5 highlight the substantial steric hindrance imparted by the 2-cycloalkyl-6-benzhydryl or 2,6-dicyclopentyl ortho-substitution pattern; distorted square pyramidal geometries are exhibited in each case. On activation with methylaluminoxane (MAO) or modified methylaluminoxane (MMAO), all the complexes (apart from Co4/MAO) were active ethylene polymerization catalysts (up to 3.70 ? 106 g PE per mol (Co) per h for Co5/MMAO), operating effectively at temperatures between 50 °C and 60 °C, producing polyethylenes with high molecular weights (up to 589.5 kg mol-1 for Co3/MAO). Furthermore, all polymers were highly linear (Tm > 130 °C) with narrow dispersities (Mw/Mn range: 2.0–3.0). The coexistence of two chain termination pathways, β-H elimination and transfer to aluminum, has been demonstrated using 13 C/1H NMR spectroscopy.
Altmetrics:


