На сайте журнала Journal of Catalysis, 2019, Volume 380, December 2019, Pages 145-152 (IF 7,723) опубликована статья с участием сотрудников Института: к.х.н. И.В. Ильиной (снс, ЛФАВ), к.х.н. Д.В. Корчагиной (снс ГЯМР), д.х.н. Ю.В. Гатилова (внс , ГРСА), д.х.н., проф. РАН К.П. Волчо (гнс ЛФАВ), д.х.н., проф. Н.Ф. Салахутдинова (зав.отделом медицинской химии)
Clay nanotubes catalyzed solvent-free synthesis of octahydro-2H-chromenols with pharmaceutical potential from (-)-isopulegol and ketones
A.Yu.Sidorenko, A.V.Kravtsova, I.V.Il'ina, J.Warna, D.V.Korchagina, Yu.V.Gatilov, K.P.Volcho, N.F.Salakhutdinov, D.Yu.Murzin, V.E.AgabekovJournal of Catalysis, Volume 380, December 2019, Pages 145-152
First published: 31 October 2019
https://doi.org/10.1016/j.jcat.2019.10.015
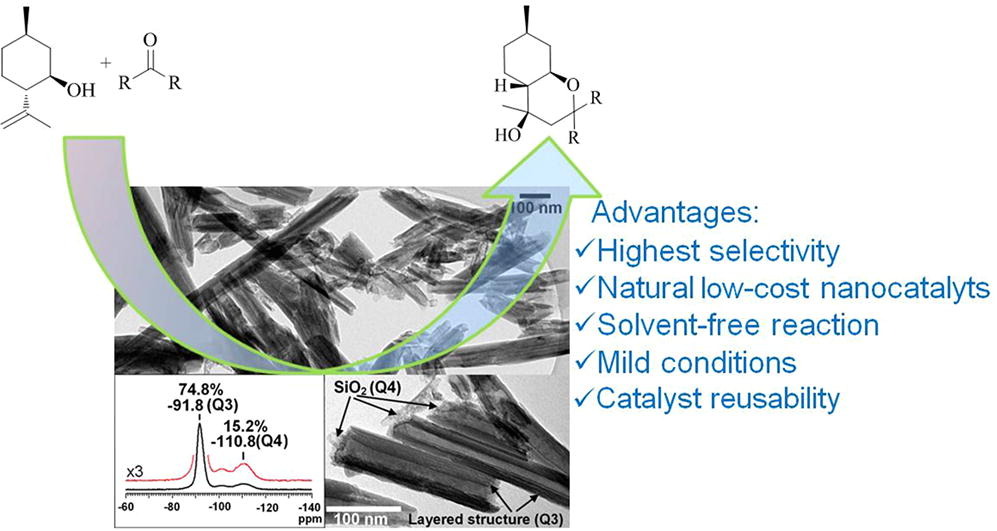
Abstract
Natural halloysite nanotubes (HNT) pretreated with hydrochloric acid were applied for the first time for selective synthesis of octahydro-2H-chromenols (as 4R- and 4S-diastereomers) using terpenoid (-)-isopulegol and ketones as reactants. The main attention was paid to the reaction with acetone, because the resulting 4R-isomer displays antiviral activity. While the drawback of the isopulegol Prins reaction with ketones is a low yield of chromenols, considerably higher values could be achieved using a halloysite catalyst under solvent-free conditions and low temperature (30 °C) giving up to 77% overall selectivity towards chromenols. The isolated yield of the acetone-derived 4R-isomer was 66%, significantly exceeding previous results. The proposed mechanism of isopulegol condensation with ketones is more complex than in the case of aldehydes. Formation of the target chromenols occurs through addition of water to the cyclic intermediate. In parallel interactions of this intermediate with isopulegol lead to stereoselective formation of an ester with the chromene structure, which after hydrolysis gives chromenols. Formation of the ether is opposite to the Prins reaction of aliphatic alcohols with carbonyl compounds, where non-cyclic acetals are typically formed. This clearly confirms that in the Prins reaction of isopulegol with carbonyl compounds, the cyclic intermediate plays a key role. A decrease in the halloysite hydration led to an increase in ether selectivity, while with the increase in the studied catalysts acidity, formation of dehydration by-products increased. A reaction scheme for the products formation was proposed and evaluated by kinetic modelling. A possibility of the catalyst reuse has been also shown. Halloysite nanotubes can be considered as highly selective, eco-friendly, low-cost and reusable catalysts for synthesis of heterocycles based on terpenoids and ketones.
Альметрики:


