В журналу Bioorganic Chemistry (IF 3,926) опубликована статья с участием сотрудников Института к.х.н. С.О. Куранова (нс, ЛНТПС ), д.х.н. О.А. Лузиной (внс ЛФАВ) и д.х.н., чл.-корр. РАН, проф. Н.Ф. Салахутдинова (зав.оделом, ОМХ, завлаб , ЛФАВ):
Exploring bulky natural and natural-like periphery in the design of p-(benzyloxy)phenylpropionic acid agonists of free fatty acid receptor 1 (GPR40)
Abstract
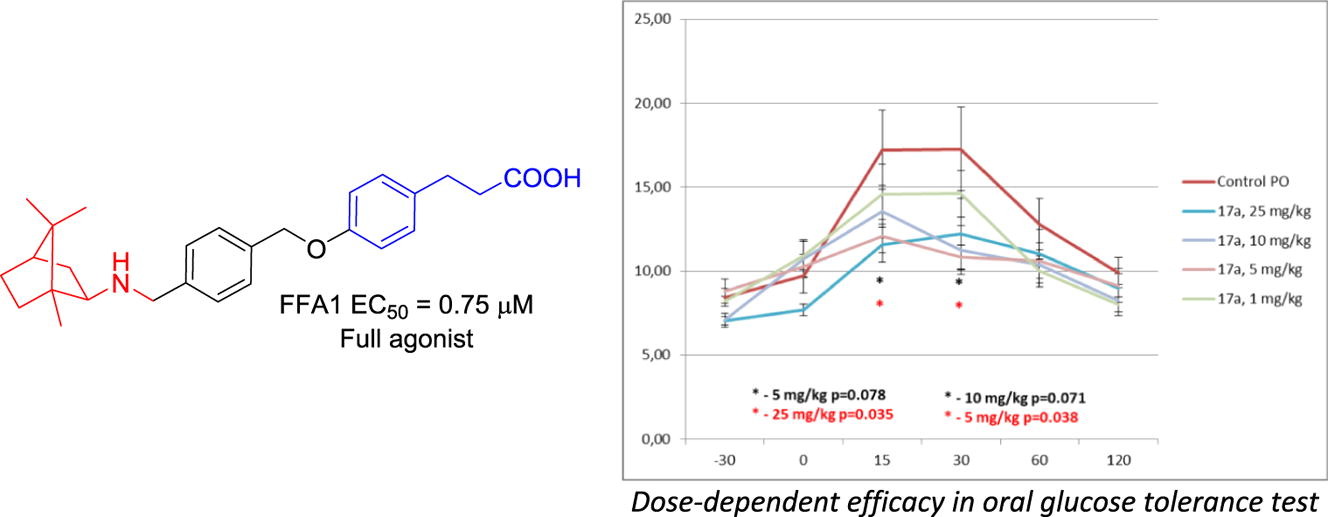
Six derivatives of 3-phenylpropionic acid bearing various natural and natural-like, spatially defined peripheral motifs have been synthesized and evaluated in vitro for free fatty acid receptor 1 (FFA1) activation. Two frontrunner compounds (bearing a bornyl and cytosine groups) were evaluated in an oral glucose tolerance test in mice where both demonstrated the ability to sustain blood glucose levels following a glucose challenge. The bornyl compound displayed a somewhat superior, dose-dependent efficacy and, therefore, can be regarded as a lead compounds for further development as a therapeutic agent for type 2 diabetes mellitus. Its high affinity to FFA1 was rationalized by docking experiments.
Альметрики:


