В журнале Journal of Natural Products (IF=3.779) опубликована статья с участием сотрудников Института: чл.-корр. РАН, д.х.н., проф. Н. Ф. Салахутдинова (рук. отдела ОМХ, завлаб ЛФАВ), д.х.н. О.И. Лузиной (внс, ЛФАВ), А.С. Филимонова (мнс, ЛНТПС)
Usnic Acid Conjugates with Monoterpenoids as Potent Tyrosyl-DNA Phosphodiesterase 1 Inhibitors
Olga Luzina, Alexander Filimonov, Alexandra Zakharenko, Arina Chepanova, Olga Zakharova, Ekaterina Ilina, Nadezhda Dyrkheeva, Galina Likhatskaya, Nariman Salakhutdinov, and Olga LavrikJ. Nat. Prod. 2020, 83,8,2320-2329
Publication Date:July 28, 2020
https://doi.org/10.1021/acs.jnatprod.9b01089
Abstract
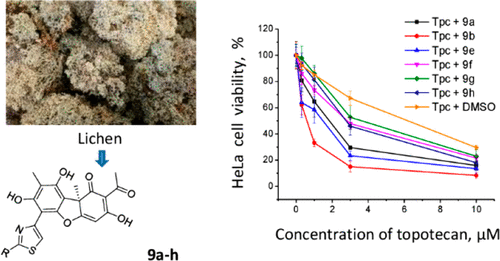
Hybrid molecules created from different pharmacophores of natural and synthetic equivalents are successfully used in pharmaceutical practice. One promising target for anticancer therapy is tyrosyl-DNA phosphodiesterase 1 (Tdp1) because it can repair DNA lesions caused by DNA-topoisomerase 1 (Top1) inhibitors, resulting in drug resistance. In this study, new hybrid compounds were synthesized by combining the pharmacophoric moiety of a set of natural compounds with inhibitory properties against Tdp1, particularly, phenolic usnic acid and a set of different monoterpenoid fragments. These fragments were connected through a hydrazinothiazole linker. The inhibitory properties of the new compounds mainly depended on the structure of the terpenoid moieties. The two most potent compounds, 9a and 9b, were synthesized from citral and citronellal, which contain acyclic fragments with IC50 values in the range of 10–16 nM. Some synthesized derivatives showed low cytotoxicity against HeLa cells and increased the effect of the Top1 inhibitor topotecan in vitro by three to seven times. These derivatives may be considered as potential agents for the development of anticancer therapies when combined with Top1 inhibitors.
Альметрики:


