The article with participation of NIOCh's researchers is published in
International Journal of Pharmaceutics (IF 5,875) :
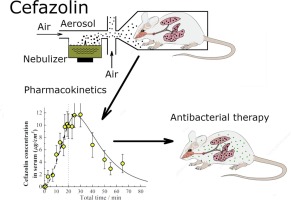
Aerosol Inhalation Delivery of Cefazolin in Mice: Pharmacokinetic Measurements and Antibacterial Effect
Abstract
Aerosol inhalation delivery of cefazolin, a broad-spectrum first-generation cephalosporin antibiotic, was investigated. Inhalation system based on ultrasonic nebulizer was developed for the generation of dry cefazolin aerosol within mean particle diameter range 0.5–3.0 μm and mass concentration 0.01–3 μg/cm3. Pharmacokinetic measurements were carried out for the aerosolized form of cefazolin delivered in mice using nose-only chamber. Cefazolin concentrations in blood serum and in the lungs of mice were measured as a function of time by means of high performance liquid chromatography. Body-delivered dose depending on particle size, concentration and inhalation time as well as other pharmacokinetic parameters were determined. The antibacterial effect of aerosolized cefazolin was assessed through the aerosol inhalation treatment of mice infected with Klebsiella pneumoniae. Survival rate for infected mice after the treatment with cefazolin aerosol demonstrated high antibacterial efficiency of the inhalation delivery of cefazolin in comparison with intraperitoneal delivery.
Альметрики:


