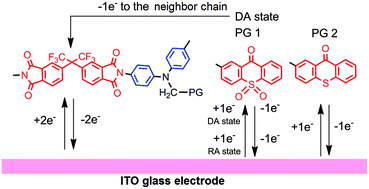
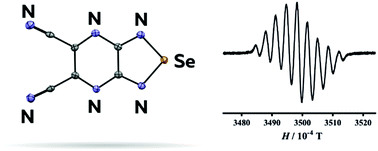
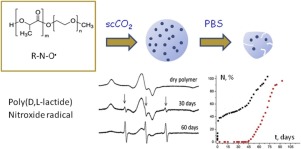
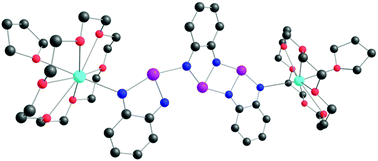
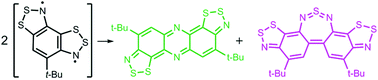
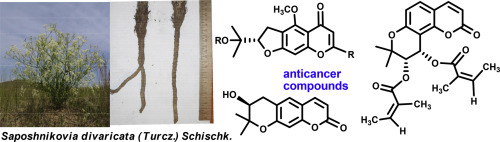
В журнале European Journal of Organic Chemistry (IF 3.029) опубликована статья с участием сотрудников Института: к.х.н. С.И. Живетьевой (нс ЛИНИРР), д.х.н. И.Ю. Багрянской (внс, руководитель группы ГРСА), к.х.н. М.С. Казанцева (снс, ЛОЭ) и д.х.н. Е.В. Третьякова (зам. директора по науке, зав.лаб ЛИНИРР)
Preparation of Multi‐Spin Systems: A Case Study of Tolane‐Bridged Verdazyl‐Based Hetero‐Diradicals
Darya E. Votkina, Pavel V. Petunin, Svetlana I. Zhivetyeva, Irina Yu. Bagryanskaya, Mikhail N. Uvarov, Maxim S. Kazantsev, Marina E. Trusova, Evgeny V. Tretyakov, Pavel S. Postnikov
European Journal of Organic Chemistry, Volume 2020, Issue13, April 7, 2020 Pages 1996-2004
First published:04 March 2020
DOI:https://doi.org/10.1002/ejoc.202000044
Hot Topic: C-C Coupling
Abstract
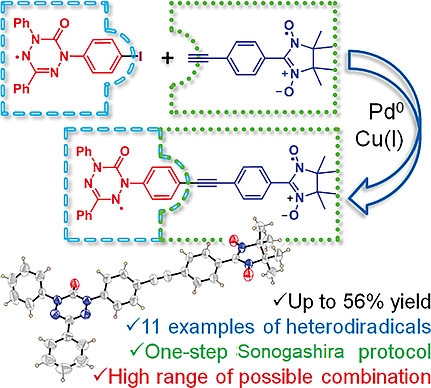
Sonogashira coupling was investigated as a promising strategy for the synthesis of a multi‐spin system based on ‘Kuhn'‐verdazyls, oxoverdazyls, and nitronyl‐nitroxides. Eleven hetero‐diradicals were isolated in up to 56 % yields by a one‐step protocol. Oxoverdazyl radicals had the highest reactivity. This research opens access to simple design and investigation of conjugated diradicals.
Abstract
Iodine‐ and ethynyl‐containing 'Kuhn'‐verdazyls, oxoverdazyls, and nitronyl nitroxides were investigated as building blocks for the preparation of multi‐spin systems via the Sonogashira reaction, and, as a result, eleven diradicals were prepared with fair yields. The reactivity of the building blocks indicates that oxoverdazyl iodides are effective starting components for the synthesis of diradicals via the Sonogashira coupling. The described one‐step protocol allows combining different spin units, thereby facilitating the design of tolane‐bridged diradicals and screening of their properties. The novel compounds were characterized by cyclic voltammetry, UV/Vis and electron spin resonance (ESR) spectroscopy. Although the electrochemical investigation and electronic spectra showed a negligible influence of radical moieties on each other, ESR data revealed a strong exchange interaction between two unpaired electrons. The prepared verdazyl‐nitronylnitroxide diradicals have high stability at storage and hold promise for further investigation and application.
Альметрики:







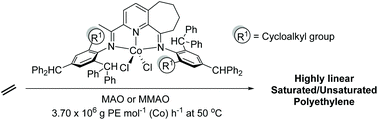
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CMe}-9-{N(Ar)}C10H10N]CoCl2 (Ar = 2-(C5H9) -4,6-(CHPh2)2C6H2 Co1, 2-(C6H11)-4,6-(CHPh2)2C6H2 Co2, 2-(C8H15)-4,6-(CHPh2) 2C6H2 Co3, 2-(C12H23)-4,6-(CHPh2 )2C6H2 Co4, 2,6-(C5H9)2 -4-(CHPh2)C6H2 Co5). All five complexes have been characterized by a combination of FT-IR spectroscopy, elemental analysis and single crystal X-ray diffraction. The molecular structures of Co1, Co3 and Co5 highlight the substantial steric hindrance imparted by the 2-cycloalkyl-6-benzhydryl or 2,6-dicyclopentyl ortho-substitution pattern; distorted square pyramidal geometries are exhibited in each case. On activation with methylaluminoxane (MAO) or modified methylaluminoxane (MMAO), all the complexes (apart from Co4/MAO) were active ethylene polymerization catalysts (up to 3.70 ? 106 g PE per mol (Co) per h for Co5/MMAO), operating effectively at temperatures between 50 °C and 60 °C, producing polyethylenes with high molecular weights (up to 589.5 kg mol-1 for Co3/MAO). Furthermore, all polymers were highly linear (Tm > 130 °C) with narrow dispersities (Mw/Mn range: 2.0–3.0). The coexistence of two chain termination pathways, β-H elimination and transfer to aluminum, has been demonstrated using 13 C/1H NMR spectroscopy.
CMe}-9-{N(Ar)}C10H10N]CoCl2 (Ar = 2-(C5H9) -4,6-(CHPh2)2C6H2 Co1, 2-(C6H11)-4,6-(CHPh2)2C6H2 Co2, 2-(C8H15)-4,6-(CHPh2) 2C6H2 Co3, 2-(C12H23)-4,6-(CHPh2 )2C6H2 Co4, 2,6-(C5H9)2 -4-(CHPh2)C6H2 Co5). All five complexes have been characterized by a combination of FT-IR spectroscopy, elemental analysis and single crystal X-ray diffraction. The molecular structures of Co1, Co3 and Co5 highlight the substantial steric hindrance imparted by the 2-cycloalkyl-6-benzhydryl or 2,6-dicyclopentyl ortho-substitution pattern; distorted square pyramidal geometries are exhibited in each case. On activation with methylaluminoxane (MAO) or modified methylaluminoxane (MMAO), all the complexes (apart from Co4/MAO) were active ethylene polymerization catalysts (up to 3.70 ? 106 g PE per mol (Co) per h for Co5/MMAO), operating effectively at temperatures between 50 °C and 60 °C, producing polyethylenes with high molecular weights (up to 589.5 kg mol-1 for Co3/MAO). Furthermore, all polymers were highly linear (Tm > 130 °C) with narrow dispersities (Mw/Mn range: 2.0–3.0). The coexistence of two chain termination pathways, β-H elimination and transfer to aluminum, has been demonstrated using 13 C/1H NMR spectroscopy.