В журнале Physical Chemistry Chemical Physics, (IF 3,945) опубликована статья с участием сотрудников Института: к.х.н. Л.В. Политанской (завлаб, ЛИНИРР), к.х.н. Ю.Ф. Полиенко (снс. ЛАС), к.х.н. В.М. Тормышева (снс, рук. ГИТР) и д.ф.-м.н., проф. Е.Г. Багрянской (Директор НИОХ)
Fullerene-based triplet spin labels: methodology aspects for pulsed dipolar EPR spectroscopy
Ivan O. Timofeev, Larisa V. Politanskaya, Evgeny V. Tretyakov, Yuliya F. Polienko, Victor M. Tormyshev, Elena G. Bagryanskaya, Olesya A. Krumkacheva and Matvey V. Fedin
Phys. Chem. Chem. Phys., 2022, 24 (7), 4475-4484,First published on 19 Jan 2022
doi: 10.1039/D1CP05545C
Abstract
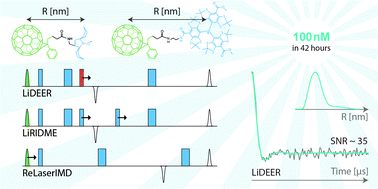
Triplet states of photoexcited organic molecules are promising spin labels with advanced spectroscopic properties for pulsed dipolar electron paramagnetic resonance (PD EPR) spectroscopy. Recently proposed triplet fullerene labels have shown great potential for double electron–electron resonance (DEER) distance measurements as “observer spins” due to a high quantum yield of the triplet state, hyperpolarization and relatively narrow EPR spectra. Here, we demonstrate the applicability of fullerene labels to other PD EPR techniques, such as relaxation induced dipolar modulation enhancement (RIDME) and laser induced magnetic dipolar spectroscopy (LaserIMD). In particular, a specific contaminating signal in LaserIMD experiments was observed, explained and mitigated. Comparative analyses of the signal-to-noise (SNR) ratios were performed for all employed methods. DEER on the fullerene–triarylmethyl pair shows the best performance, which allows state-of-the-art DEER acquisition at 100 nM with a SNR of ∼35 within reasonable 42 hours.
Альметрики:









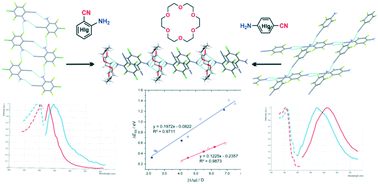
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C and N–H⋯F) by the bond with a crown oxygen atom (N–H⋯Ocr). The crystal packing rearrangement modifies the π-electron interactions between aminobenzonitrile molecules both in the type of contact (C–F⋯π, C–Cl⋯π, C
C and N–H⋯F) by the bond with a crown oxygen atom (N–H⋯Ocr). The crystal packing rearrangement modifies the π-electron interactions between aminobenzonitrile molecules both in the type of contact (C–F⋯π, C–Cl⋯π, C